Protocols for Mare Management with Cooled-Shipped or Frozen Semen
By Jos Mottershead
Mare management with cooled-shipped or frozen semen can often seem daunting but is a key element for success or failure. One can use or forgo pharmacological manipulation, and we discuss here possible protocol variables and alternatives with both methods.
Using No Pharmacological Manipulation
Obviously one major key ingredient in any breeding program from a mare owner’s point of view, is the awareness of when the mare is in estrus and the right time to breed. This is especially so in a breeding program that uses artificial insemination of cooled transported or frozen semen. This can be successfully achieved by careful monitoring with palpations and ultrasound, the “teasing” of mares, or – most effectively – a combination of all. Geldings may be used to tease mares, but if mares are usually housed with the geldings or in an adjacent pasture, it may be necessary to remove one or other from the area for a major portion of the day, and monitor the response when the animal is re-introduced.
One common error with mare owners who monitor their mares without the use of a stallion to tease, is the belief that when they first see the mare showing signs of estrus she is just entering the estrus phase of the cycle. Often though, the mare is in an advanced state of estrus when she finally “shows” – her response is a desperate plea for the attention of a stallion NOW! This response may not be seen until the mare is very close to ovulation. Misinterpretation of this being the start of estrus will usually result in missing the ovulation.
If one has a mare confirmed to be just entering estrus, it is usually safe to wait until about day three of display before a veterinarian is needed to evaluate her ovarian state. This is assuming that the mare is one that shows a “normal” estrus duration of 5 or 6 days. Most mares will ovulate 24 to 48 hours prior to the end of estrus as evidenced by “standing heat”. A mare that is examined at about day 3 of estrus will be “mid-estrus”, and will be likely to have a dominant follicle in excess of 3½ centimeters in average diameter. At this time an ultrasound review of follicular size and activity, level of uterine edema, determination of presence of fluid in the uterus (and treatment if present), and the observation and recordation for future reference of the position and size of any uterine cysts should be carried out. If the mare has not had a uterine swab or low-volume lavage sample taken for cytogenic and bacterial evaluation on a previous cycle, this may be taken now. It should be noted that the results of a culture may not be returned in time to breed on the same cycle, but a cytology smear (which takes only minutes to obtain and evaluate) looking for the presence of neutrophils can be used to suggest the possible presence or absence of a pathogen. If neutrophils are present, then a culture of the swab should be prepared and read, with a test of the resulting growth for sensitivity towards various antibiotics performed. A cytology smear is an essential diagnostic, and the results of a uterine swab culture alone should be viewed with caution or even ignored if they are not supported by the findings of a cytology smear.
If uterine fluid is seen at this stage per ultrasound, providing neutrophils have not been detected in the cytology smear, (which would indicate inflammatory response, and therefore possible infection), the use of oxytocin may be adequate to clear the fluid. This can be coupled with systemic injectable estrogen, or topical treatment of the cervix with Misoprostal or Buscopan, all to encourage relaxation of the cervix which aids in the expulsion of the fluid. Poor uterine tone may be aided by a similar treatment, or with the use of oxytocin alone. If the measured fluid depth exceeds 2 cm, uterine lavage with either sterile saline or Lactated Ringer’s solution coupled with oxytocin treatment is indicated prior to commencing breeding.
If all appears to be in good shape and a follicle in the 3½ cm or larger range is found, coupled with uterine edema the mare can be “followed” using rectal palpation or ultrasound. Repeated ultrasounds can increase breeding costs significantly, so a combination of palpations and ultrasound examinations may be both cost-effective and efficacious, althought these days it is more common to see ultrasound alone used. The visible presence of uterine edema per ultrasound is essential, as that indicates true estrus rather than a mid-cycle follicle. Breeding in the absence of uterine edema is typically going to be detrimental to the reproductive health of the mare. Diestrus follicles can and do occur, however the mare will have elevated levels of progesterone – which is normal during diestrus – so uterine infection will be a common sequela to such breeding, coupled with a lack of pregnancy. The required number and frequency of checks will vary depending upon the results of the initial ultrasound examination – and often the value of the semen! If the follicle size is tending towards smaller at the initial evaluation, it will probably be safe to wait up to 2 days before further evaluation. If the follicle is larger, noted to be suggestive of being closer to ovulation, or the mare has a history of ovulation on smaller follicles, then the next check should be carried out much sooner. Evaluation of uterine edema levels can also be a useful indicator of impending ovulation, as such edema will typically peak around 36-24 hours prior to ovulation in the reproductively healthy mare. In mares with delayed uterine clearance issues or with a uterine infection, the edema may persist through to or after ovulation.
When imminent ovulation is anticipated as a result of ultrasound evaluation showing a change in follicular shape, thickening of the follicular wall, and reduction in uterine edema, or the “softening” of a large follicle per rectal palpation, checks should be carried out as often as every 6 hours. In some cases, practitioners may even choose to check as often as every 4 hours, while other research has suggested that increasing the interval to as much as every 12 hours will still produce the same pregnancy rate1. The accepted dogma for the timing of artificial insemination of frozen semen however is no more than 12 hours prior to, or 6 hours after ovulation. The ideal time for insemination is right at the point of ovulation, or within the 6 hours preceding, although inseminating prior to ovulation does carry the risk of premature use and the mare not ovulating even though the only available dose of semen was used! There is a greater incidence of early embryonic loss in mares inseminated greater than 12 hours after ovulation, although this time frame may be extended to 16 hours if good post-breeding management is used, including use of oxytocin and uterine lavage1.
The use of ultrasound the day after insemination (assuming one has determined a timely ovulation) is valuable to evaluate fluid build up, if present. Some mares will show a greater inflammatory response and associated fluid than others. It is particularly noted in older mares, and is believed to be more of an indication of the mare’s inability to clear the fluid, rather than an excessive response itself. The actual causative agent of the response has been shown to not be the egg yolk or glycerol in the extender as was once thought, but the sperm themselves.
 If fluid is present post breeding, the use of an oxytocin protocol can be beneficial. Multiple treatments of 10-20 IU, 6 hours apart can be carried out until as late as 3½ days post-ovulation. Some like to routinely use 10-20 IU of oxytocin 4 hours after breeding with frozen or cooled transported semen, as the natural release of oxytocin that occurs during live cover is reduced with AI. The use of prostaglandin F2α or one of its synthetic analogues such as cloprostenol (“Estrumate” – Schering-Plough Corporation Kenilworth, NJ USA) – which has a longer half-life and can therefore be used every 12 hours (rather than every six hours, such as oxytocin requires) to aid post-breeding clearance has been shown to interfere with the successful formation of a fully functional corpus luteum. Its use is therefore contra-indicated after 12 hours post-ovulation, although prior to that it may be of value.
If fluid is present post breeding, the use of an oxytocin protocol can be beneficial. Multiple treatments of 10-20 IU, 6 hours apart can be carried out until as late as 3½ days post-ovulation. Some like to routinely use 10-20 IU of oxytocin 4 hours after breeding with frozen or cooled transported semen, as the natural release of oxytocin that occurs during live cover is reduced with AI. The use of prostaglandin F2α or one of its synthetic analogues such as cloprostenol (“Estrumate” – Schering-Plough Corporation Kenilworth, NJ USA) – which has a longer half-life and can therefore be used every 12 hours (rather than every six hours, such as oxytocin requires) to aid post-breeding clearance has been shown to interfere with the successful formation of a fully functional corpus luteum. Its use is therefore contra-indicated after 12 hours post-ovulation, although prior to that it may be of value.
Using Pharmacological Manipulation
Prostaglandin F2 alpha (PGF2α)
The use of prostaglandin (PGF2α) – in North America found in the product names “Lutalyse” or “Prostin F2 Alpha” (Pharmacia & Upjohn Company, Kalamazoo, MI), or “ProstaMate” (Phoenix Pharmaceutical, Inc St. Joseph, MO) – is probably the most common form of hormonal manipulation of the mare used to promote estrus (often referred to as “short cycling”). The problem with the use of this hormone is that it will only be reliable if given at a certain point in the mare’s cycle. There must be a receptive Corpus Luteum (CL) for the PGF2α to act upon, so use within about 4-5 days after an ovulation or in an anestrus mare will not result in timely production of estrus. One can simply rely on luck and external observation to ensure that the hormone is given a minimum of 6 days after the mare has ovulated, but even assuming that you are fortunate enough to be able to achieve this, there is still the possibility that the mare will have produced a “mid cycle follicle”. If the PGF2α is given with a large active follicle present, there is the possibility that the mare will enter estrus and ovulate within 3 days or sooner after dosage, with lowered fertility2. We discuss the pitfalls of prostaglandin in our article “I gave my mare prostaglandin but it didn’t work“.
It should be noted that a CL may appear to be visible per ultrasound, but be a remnant from a prior cycle and no longer secreting progesterone. In order to ensure there is a functional CL present, it is possible to carry out a blood-progesterone assay. If progesterone is present in levels of 1 ng/ml or greater, this indicates that there is a functional CL present. Levels of 1 to 3 ng/ml may indicate that the mare is either entering or leaving estrus, in estrus, or in a state of anestrus, and the testing should be repeated in 2-3 days. If the progesterone levels have increased at the time of the subsequent testing, it is indicative of leaving estrus, and the mare may well already have – or will shortly have – a CL receptive to prostaglandin. If the progesterone level has dropped on the subsequent test, she is entering estrus (or more rarely anestrus), and normal monitoring procedures may be followed. Onset of physiological estrus is normally anticipated 3 to 5 days following the administering of PGF2α at the right stage in an estrous cycle if there is no active mid-cycle follicle present.
The success rate of prostaglandin use to synchronize mares is limited, although a reasonable degree of synchronicity is seen if mares are given two full doses of prostaglandin 14 days apart. With that technique, by the time of the second dose being administered, both mares should have a receptive CL present. Although not researched or demonstrated, use of the two micro-doses of PGF2α on days 0 and 1 and again on days 13 and 14 should produce the same effect without the unpleasant side-effects (see below).
A common cause for concern connected with the use of prostaglandin to short cycle mares are the potentially dramatic side effects. Transient sweating and colic-like symptoms are frequently seen commencing 5 to 10 minutes after dosage. These symptoms have usually disappeared an hour later, but often not before causing significant alarm for the mare owner! The cause of the side effects is stimulation of adrenoreceptors on the sweat gland cells3. Cramping may also occur owing to contraction of smooth muscle of the uterus. It is extremely rare that these side-effects cause significant problems, although if they do, most commonly the affected horse has reduced water consumption, resulting in a full-blown colic. The use of cloprostenol (“Estrumate” – Schering-Plough Corporation, Kenilworth, NJ) – a prostaglandin analogue – rather than native prostaglandin F2α may avoid these side-effect, although not in all animals. It should be noted that Estrumate is not labelled for use in the equine in the USA, but is commonly used off-label for this purpose. It has been shown that the use of one-tenth of the normal dose of prostaglandin or its analogue given on two consecutive days at least 6 days after the previous ovulation will result in a return to estrus in the same 3-5 day interval as the full dose, but without the unpleasant side effects for the mare4. It is believed that this lack of side effects is as a result of the compounding of the low dose more closely mimicking the natural release of prostaglandin.
Progesterone or Altrenogest
Progesterone – “P4”, an endogenous steroidal hormone – is available in the USA as a compounded product from a variety of compounding pharmacies. The synthetic progestin Altrenogest is commercially produced as RegumateTM (Merck Animal Health – Madison, NJ, USA) or in a compounded injectable biorelease 12 or 30 day formulation (BET Pharm, Lexington, KY USA). One of the most important points for consideration in the use of P4 or progestins is that although they will suppress estrus, they will not necessarily suppress ovulation. This means that their use is not a guaranteed method of promoting timely estrus. If the mare is producing what in effect becomes a mid-cycle ovulation during treatment, it may throw her natural cyclical activity into an erratic state. Consequently, the mare may not come into estrus at the anticipated “normal” 3 days post cessation of the treatment. We discuss the pitfalls of progestin use in our article “I gave my mare progesterone (or Regumate) but it didn’t work“.
Progesterone and Estradiol (“P&E”):
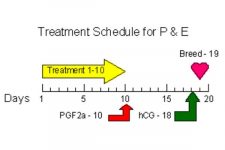
The administering of a combination of progesterone and estradiol – specifically estradiol 17β – produces a high degree of control over the mares estrus cycle5,6,7. The estradiol (estrogen) portion of the mixture suppresses follicular development and ovulation of any follicle present, allowing the progesterone to achieve the effect that is desired – but often doesn’t occur – by its use alone. There is currently no “brand name” product combining these 2 hormones, but it can be obtained from some veterinary compounding pharmacies. It is available in an injectable form that requires injections once a day for 10 days or in a single-injection biorelease formulation (BET Pharm, Lexington, KY USA). Prostaglandin should be administered in conjunction on the 10th day, or if using the reduced prostaglandin dosage, we have found that it may be given on the 9th and 10th days, resulting in no unpleasant side-effects, and no negative impact on the P&E protocol. Ovulation will occur in over 80% of mares, when combined with an ovulation promoter, 9 days after the last administration of the daily P&E dosage, or approximately 19 days after the treatment with the biorelease formulation. The use of this hormonal combination is discussed at greater length in an article elsewhere on this web site.
While the drawback of 10 daily injections may be overcome by the use of a biorelease formulation of P&E (BET Pharm, Lexington, KY, USA) as noted, the timing of the resulting ovulation may not be quite as accurate as that of the daily dosing system, however this can often be overcome with careful monitoring and timely use of an ovulation promoter. Daily oral dosing has been attempted, but results have been unpredictable. The use of a vaginal sponge, or dosing directly with the daily treatment topically in the vagina has been seen to produce reliable results8, although a transient vaginitis may be present that resolves spontaneously before the onset of estrus. Research suggests that the biorelease formulation of altrenogest (BET Pharm, Lexington, KY, USA) may produce similarly accurate timing of ovulation as the biorelease P&E9.
Ovulatory Promoters:
Promoting ovulation may be beneficial, offering a more accurate prediction of the timing of the ovulation and a reduction in necessary monitoring. Commonly used ovulation stimulatory agents include hCG; synthetic forms of GnRH – deslorelin, histrelin or buserelin; and recombinant LH.
Human Chorionic Gonadotropin (hCG)
Human chorionic gonadotropin (hCG) is commercially available as Chorulon in North America (Merck Animal Health – Madison, NJ, USA). In order for hCG to produce reliable timing of ovulation, it must be given when there is at least a 35 mm follicle present. The usual dosage for a 1,000-1,500 lb. horse is considered to be anywhere between 1,500 and 3,300 IU, although 1,500 IU has been shown to be an adequate dose10. Overdosing – at 5,000 IU or greater – has been seen to potentially suppress ovulation rather than promote it. It is worthy of note that hCG is commonly packaged in 5,000 and 10,000 IU vials, representing multiple doses, and rather than wasting the unused portion, freezing individual doses in suitable-sized syringes for use within the same breeding season makes the product particularly cost-effective. Repeated use of hCG in the same mare within the breeding season has been suggested as resulting in a reduction in reliability of ovulation promotion, especially in older mares11, although this has been debated10, and clinical evidence strongly suggests an individual response.
GnRH and its analogues
The synthetic GnRH deslorelin is presented in a commercially prepared time-release implant in the product “Ovuplant” (Wyeth Animal Health, Guelph, ON, Canada; Peptech Animal Health, Sydney, NSW, Australia) but shortly after introduction to the US market it became apparent that in the event of failure of pregnancy establishment or the flusihng of an embryo, return to subsequent estrus – especially if PGF2α was used in an attempt to promote estrus once pregnancy establishment failure was identified or an embryo flushed – could be seriously delayed12. Despite developments of techniques wherein the delay was eliminated, involving removal of the implant 48 hours after implantation13, the product was removed from the US market and remains unavailable in the USA, although it is still used in other countries.
Following the removal of Ovuplant from the US market, compounded Deslorelin became available and a biorelease formulation made by BET Pharm (Lexington, KY, USA) was particularly reliable. This however was also removed from the market following the FDA approval of a new Deslorelin product “Sucromate” (it is illegal for compounding pharmacies to produce compounds of drugs that are commercially available and which have undergone FDA testing and approval).
A compounded time-release formulation of another GnRH analogue “Histrelin” is available (BET Pharm, Lexington, KY, USA) and has been shown to be efficacious in promotion of ovulation within 48 hours if given to an estrus mare in the presence of a 30 mm follicle, with possibly improved conception rates when compared to other ovulation induction agents14.
In countries other than the US, the GnRH analogue “Buserelin” may be available as the commercial product “Suprefact” (Sanofi-Aventis, Reading, Berkshire, UK), which has been shown to be effective at promoting ovulation and cheap owing to the low dose required15.
Monitoring of the Mare Undergoing Pharmacological Manipulation
If there is every reason to believe that the mare has entered estrus at the anticipated point, then the same protocol as is followed for the non-manipulated mare may be followed for the mare that has undergone treatment with hormones. Where the problems arise are in those mares that do not respond to hormone use as anticipated, often as a result of the hormones being administered at the wrong point in the cycle (especially PGF2α), or the presence of a large mid-cycle follicle. The use of “P&E” is strongly recommended to avoid these misadventures if careful pre-treatment monitoring with ultrasound is not an option.
With the use of “P&E”, a high percentage of mares (in the region of 80%) will enter timely estrus and then ovulate 9 days after the last dose of the daily hormones – or around 19 days after treatment with the biorelease formulation – if an ovulation promoting agent is given on the morning of day 8/18 (respectively) if the follicle then present is between 35 and 40 mm in size. Larger follicles may result in a more rapid ovulation. Administering hCG when follicles are smaller than 35 mm, or deslorelin or histrelin when follicles are smaller than 30 mm, will have an unreliable or no effect on promotion of ovulation.
The mare should receive an ultrasound examination 5 or 6 days after the administering of the last daily dose of P&E, or around 15 days after treatment with the single biorelease formulation. If not previously taken, a uterine swab sample may be obtained at this point, for the purposes described above, however as noted, culture results may not be returned in time to breed. If a follicle of approximately 30 to 40 mm is present at this stage (depending upon promoter being used), the ovulation promoting agent may be administered with awareness that the ovulation may occur slightly earlier than 18/19 days after initial treatment. Palpations or ultrasounds should be increased to as often as every 6 hours in the period commencing 24 hours after use of the ovulation promoter. If a smaller follicle is present, a repeat ultrasound examination in 24-48 hours is recommended.
It is worth noting that if one is not fortunate enough to observe such items as the “pear shaping” of a follicle, thickening of the follicular wall, and/or the peaking and reduction of uterine edema, it may not be easy to predict the impending ovulation using ultrasound alone. Rectal palpation to assess the “softness” of the follicle coupled with the ultrasound assists in reliability of predicting the likelihood of impending ovulation. Ultrasound remains more reliable than palpation alone to confirm ovulation did occur if a definitive ovulatory depression is not palpated. We further discuss indications of impending ovulation in our article so titled.
Pharmacological manipulation – at the very least use of an ovulatory promoting agent – also opens the door to the use of timed insemination protocols17, thereby reducing the number of ultrasounds and/or palpations required. This can save on breeding costs significantly, although the need for multiple insemination doses may become a factor if buying semen “by the dose”.
Adaptation for Cooled (Transported) Semen Use
Very much the same protocols can be used with cooled (transported) semen, although a relaxation of timing of checks is allowed. As cooled semen will last 24-48 hours in the mare (or longer) there is no need to breed so closely to ovulation, and as a consequence the need to check the mare every 6 hours is negated. Checking every 24 or even 48 hours is adequate. Semen is typically ordered for overnight shipment when a 3½ cm follicle is identified and an ovulation promoter used to “push” the ovulation. Suitable timing of the use of that promoter is debated. If used when semen is ordered, the mare should be very close to ovulation when the semen is received 24 hours later. If however the semen is delayed or lost in transit, one will be likely to miss the ovulation with a subsequent shipment. Waiting until receipt of the semen seems more ideal, but if the mare “hangs on” before ovulating, in some cases the semen may no longer be viable and a second shipment required, or a pregnancy not established. For these reasons, the use of frozen semen – which can already be stored on-site – can sometimes be easier than cooled! Post-breeding management (as below) is the same for both cooled and frozen semen, as described.
Post-Ovulatory Monitoring
It is important to continue to assess the mare post-breeding to ensure that ovulation did indeed occur at the anticipated time if a post-ovulation breeding was not performed. Mares are notorious for proving even the experienced palpator or ultrasonographer wrong! Palpation – with the above-noted limitations – or ultrasound may be used to confirm ovulation, although palpation alone should be followed with ultrasound to evaluate the uterine environment as discussed above. If such fluid is identified, it should be treated appropriately with uterine lavage and/or oxytocin. Mares deemed susceptible for delayed uterine clearance problems bred with frozen semen may be more inclined to develop a greater post-breeding inflammatory response.
Summary:
Mare management with cooled-shipped or frozen Semen without the use of pharmacological manipulation can be successfully achieved, but may prove more difficult, costly and less successful than live cover or on-farm AI, especially in the absence of a “teaser” stallion or gelding. The level of success will depend greatly on the degree of monitoring and the experience level of those monitoring the mare. Pharmacological management if correctly performed can offer a more predictable timing for onset of estrus and the resulting ovulation in many, if not most, instances. This can make the level of success greater, and often the breeding process cheaper in the long run notwithstanding the cost of the drugs.
NOTE: The hormones used in manipulation of the mare are the same hormones that are present in humans, and they can be absorbed through the skin if spilled. All asthmatics regardless of sex and all pregnant women must be particularly careful when handling prostaglandins or prostaglandin analogues. Additionally, Progesterone, altrenogest, or Regumate must be handled extremely carefully by all women to avoid potentially significant disruption of menstrual cycles.
References
2: Lindenburg H, Koskinen E, Huhtinen M, Reilas T, Perttula H, Katila T. 2002; Influence of PG administration and follicle status on the number of conceptuses Theriogenology, 58 2-4; 571-574.
3: Jenkinson DM, Elder HY, Bovell DL. 2006. Equine sweating and anhidrosis, part 1: equine sweating. Vet Dermatol 17: 361–392
4: Wüstenhagen A, Handler J, Kindahl H and Aurich C. 2002; Luteal function and estrous cycle characteristics in mares treated with subtherapeutic doses of prostaglandin F2α Theriogenology, 58: 2-4; 537-539.
5: Loy, R.G., Pemstein, R., O’Canna, D., and Douglas, R.H. 1981; Control of ovulation in cycling mares with ovarian steroids and prostaglandin. Theriogenology 15:191-200.
6: Varner, D.D., Blanchard, T.L., and Brinsko, S.P. 1988; Estrogens, oxytocin and ergot alkaloids – Uses in reproductive management of mares. Proc. Am. Assoc. Equine. Pract., 219-241.
7: Lofstedt, R.M. 1988; Control of the estrous cycle of the mare. The Veterinary Clinics of North America – Equine Practice, 189-190.
8: Lofstedt, R.M. 2002; personal communication.
9: Burns P.J. 2007; personal communication.
10: Gastal E.L., Silva L.A., Gastal M.O., Evans M.J. 2006; Effect of different doses of hCG on diameter of the preovulatory follicle and interval to ovulation in mares; Animal Repro. Sci. 94 (2006) 186-190.
11: McCue P., Hudson J.J., Bruemmer J.E., Squires E.L. 2004; Efficacy of hCG at Inducing Ovulation: A New Look at an Old Issue; Proc. AAEP 1492:1204.
12: Johnson, C.A., Thompson, Jr. D.L., Kulinski K.M., and Guitreau A.M. 2000; Prolonged interovulatory interval and hormonal changes in mares following the use of OvuplantTM to hasten ovulation. J. Equine Vet. Sci. 20:331-336.
13: Farquhar V.J., McCue P.M., Carnevale E.M., Nett T.M, Squires E.L. 2002; Deslorelin acetate (Ovuplant) therapy in cycling mares: effect of implant removal on FSH secretion and ovarian function; Equine Veterinary Journal 34:4;417-420
14: Kiser A.M, Sudderth A.K., Brinsko S.P., Burns P.J., Douglas R.H., and Blanchard T.L. 2013; Comparison of Efficacy of Two Dose Rates of Histrelin for Inducing Ovulation in Broodmares. J. Eq. Vet. Sci. 32;4:208-210
15: Newcombe J, Cuervo-Arango J. 2017. What are the options for induction of ovulation in the mare in Europe? Buserelin as an alternative to hCG. JEVS 51:8-17
16: Niswender K., Roser J.F., Boime I., Colgin M. 2006; Induction of Ovulation in the Mare With Recombinant Equine Luteinizing Hormone; Proc. AAEP 387:388
17: Barbacini S. 2000; Management of mares for frozen semen. Proc. 14th Int. Congr. Anim. Reprod. AI; 308.
© 1998, 2002, 2007, 2014, 2021 Equine-Reproduction.com, LLC
Use of article permitted only upon receipt of required permission and with necessary accreditation.
Please contact us for further details of article use requirements.
Other conditions may apply.