Placentitis in the Mare – Diagnosis, Treatment and Outcome
By Jos Mottershead & Kathy St. Martin
Your mare is at approximately 250 days of pregnancy and one morning you notice significant mammary development. Upon closer inspection you find that milk is present and can be expelled from the udder. What to do?
Outline
Placentitis is defined as “inflammation of the placenta”, the portion primarily being affected being the allantochorion. The condition may occur in between 3% and 7% of equine pregnancies and 90% of the causative infections are due to bacteria entering the vagina, travelling up to the cervix and penetrating the cervical barrier.[1] A variety of occurrences are then possible, including separation of the placenta from the uterine lining, reduction of oxygen and nutrient transfer to the fetus, and fetal death in utero or abortion of a live, premature and/or compromised fetus.
Diagnosis
External Signs
While some mares normally develop a filled udder as much as a month before foaling, early development is commonly a sign of an issue with the pregnancy. In this latter problematical situation, if the cause is left untreated, typically an abortion will result. The premature mammary development in itself is merely an indicator of there being an issue, and may occur as a result of a variety of causes such as the mare being about to abort twins, or – most commonly – placentitis. There is no doubt that early mammary development is therefore a cause for concern and that it is necessary to determine and treat (if possible) the underlying cause.
There is a difficulty in identifying what is “premature filling” of the udder. Sometimes – such as at cited 250 days – there is no difficulty recognising the prematurity, but as the mare gets closer to her anticipated range of normal foaling dates – 320-370 days – it can become increasingly difficult to definitively identify filling as premature. Some mares may develop udder filling earlier, others closer to foaling. Maiden mares are particularly notorious for little or no development until in some cases, after foaling. It is clear therefore that “individual circumstances” are the order of the day and if the mare has foaled before a history of previous development may be a valuable indicator. As a very rough general rule of thumb, development prior to 300 days of gestation may be considered suspicious, but again it must be reinforced that there is a strong individual mare influence.
If exudate is visible either on the vulval region or pooled in the vagina, a swab sample may be taken and cultured with a view to gaining an impression of any pathogen involved. The result could lead to determination of a suitable antibiotic treatment protocol, but it must be remembered that the vagina is commonly inhabited by organisms which may or may not be associated with the placentitis. That factor could result in a return of an inaccurate diagnosis of the causative agent, although inclusion of treatment of any identified likely organisms in a treatment protocol is more likely to prove beneficial than unnecessary.
Palpation and Ultrasound
The initial step is going to be rectal palpation and ultrasound. The palpation is valuable for determining if the fetus is indeed still alive – one would not want to attempt to maintain a deceased fetus in utero – although in rare instances it may also be able to determine the presence of twins. Do bear in mind that this latter aspect is not going to be easy, and unless something definitive is clearly palpable such as two adjacent heads, the result will often be inconclusive as far as determining twin presence (or absence). It should also be borne in mind that the fetus during late pregnancy may have periods of dormancy extending to as much as an hour in duration, although usually less. It may be necessary to poke or prod the fetus during a prolonged – up to 15 continuous minutes – period of rectal examination in an attempt to elicit response. A repeat palpation may also be required later to definitively rule out fetal activity before presuming it to be dead if no movement is felt during the initial palpation. If a fetus is found to be lacking tone – (“limp”) upon palpation – a compromised foal should be anticipated. Doppler ultrasound may also be used to evaluate fetal heartbeat if within reach of the ultrasonographer.
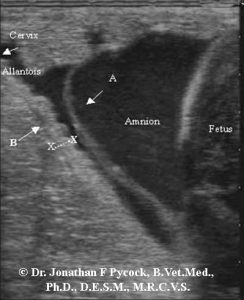
Regular ultrasound is used to evaluate placental condition – in particular “CTUP” (combined thickness of uterus and placenta also sometimes referred to as “CUPT” – combined uterine and placental thickness), the degree of attachment or separation of the placenta, and the condition of the fluids surrounding the fetus. Normal CTUP measurements vary during the pregnancy and in affected animals: “Measurement of the CTUP below 5 mm may be difficult to measure accurately at farms, and the present study suggests that a CTUP of up to 7 mm prior to day 300 of gestation may be considered normal under field conditions. A CTUP above 2 SD [standard deviations] of the established normal range after 271 days of gestation suggests placental failure and pending abortion (8 mm between days 271 and 300; 10 mm between days 301 and 330; and 12 mm after day 330). A CTUP above 10 SD of the normal range prior to 270 days of gestation also indicates pending abortion. A CTUP slightly above normal but below that for placentitis should be monitored closely until placental disease can be ruled out, based on return to a normal CTUP or absence of any clinical evidence of disease.” [2]. It should be noted that those measurements were established in Quarter Horses, and that other types of full-size horse may have a slightly greater thickness (e.g. Warmbloods) or be lesser in ponies. The majority of mares will have a CTUP ≤12 mm at the time of foaling and a CTUP >15 mm in horse mares and >12 mm in pony mares after 310 days of gestation is associated with placental malfunction.[3] Some more recent research has however questioned the reliability of CTUP measurements as an indicator of placental health during the last 30 days of pregnancy. [4] Other clinical signs of placentitis may – but do not necessarily – include discharge from the vulva (via the cervix) as previously noted, and/or cervical dilation. Ultrasonic evaluation of the allantoic fluid is also of use, with echoic particulate presence within the fluid being suggestive of there being an issue.
 CTUP measurement is performed on the caudal allantochorion within 1 to 2 inches of the cervical region, cranial of the cervical-placental junction with the probe being moved laterally until the middle branch of the uterine artery is visualized along the ventral aspect of the uterine body. The measurement is then taken at the caudal, ventral edge of the uterus between the middle branch of the uterine artery and the allantoic fluid.[3] The CTUP should not be measured along the dorsal border of the uterus as the placenta is normally thicker and edematous in that area, particularly during later pregnancy. Care must be taken not to include the amniotic membranes in the measurement, which in some cases may be closely positioned. In addition to CTUP measurement, ultrasonographic evaluation may identify placental separation from the endometrial lining, or even areas of purulent material between the two. It is important to note that the CTUP measurement will be invalid in the case of separation or if there is purulent material between the allantois and the uterine wall. Care must also be taken to not mistake the amnion for allantoic separation. Other limitations of ultrasonography in this context include minimal accessible evaluation location, and the possible need for repeated evaluation.
CTUP measurement is performed on the caudal allantochorion within 1 to 2 inches of the cervical region, cranial of the cervical-placental junction with the probe being moved laterally until the middle branch of the uterine artery is visualized along the ventral aspect of the uterine body. The measurement is then taken at the caudal, ventral edge of the uterus between the middle branch of the uterine artery and the allantoic fluid.[3] The CTUP should not be measured along the dorsal border of the uterus as the placenta is normally thicker and edematous in that area, particularly during later pregnancy. Care must be taken not to include the amniotic membranes in the measurement, which in some cases may be closely positioned. In addition to CTUP measurement, ultrasonographic evaluation may identify placental separation from the endometrial lining, or even areas of purulent material between the two. It is important to note that the CTUP measurement will be invalid in the case of separation or if there is purulent material between the allantois and the uterine wall. Care must also be taken to not mistake the amnion for allantoic separation. Other limitations of ultrasonography in this context include minimal accessible evaluation location, and the possible need for repeated evaluation.
Endocrinology
In the healthy pregnant mare, estrogen levels rise to a peak at around 210 days. Measurement of levels between 150 and 280 days of pregnancy are considered to be indicative of fetal well-being. Concentrations in that period >1,000 pg/ml are normal, while levels <1,000 pg/ml are thought to be indicative of fetal stress or lack of viability. Levels prior to 300 days of gestation between 500 and 800 pg/ml may indicate a compromising of the fetus, while levels <500 pg/ml are strongly suggestive of either severe compromise or death of the fetus. Progestogen levels – note “progestogen” not “progesterone”, levels of which drop to base-line at around 100-150 days – remain relatively low throughout later normal pregnancy until about 15-21 days before foaling, at which point levels rise dramatically, only to drop equally dramatically about 24 hours prior to parturition. Douglas showed that progestogen levels range between 4 and 10 ng/ml until day 300, reaching a peak mean of approximately 26 ng/ml by day 340. He also observed that is important to recognize that between days 150 and 280 of pregnancy, placentitis usually results in higher progestogens and lower estrogens when compared to normal mares. Thus, a single serum hormonal assay for these two hormones can add valuable information to enable the attending veterinarian to confirm the presence of placentitis and indicate that both antibiotic and hormonal therapy may be needed [5], while other practitioners suggest a series of three samplings taken over a 48-72 hour period. This same source points out that the estrogen assay will be unreliable after about 300 days of gestational duration owing to the naturally dropping levels at that point.[1]
There is a possible blood test for placentitis with evaluation of serum amyloid A (SAA).[6] Inflammation results in elevation of serum concentrations of acute phase proteins, of which SAA is the major one present in the horse. Caution must be observed with testing for SAA however, as an elevated level will not be specific to placentitis, but will merely indicate a general level of inflammation present somewhere in the body.
Treatment
In the multi-faceted treatment protocol for placentitis, there are several aims: control infection, reduce the inflammation, possibly increase bloodflow to the uterus, and encourage myometrial quiescence:
Infection:
Containing and controlling the infection is usually achieved with antibiotic therapy. Owing to the infectious agent being located within the uterine environment, as noted swabbing of the vagina may produce unreliable results for identifying the causative pathogen with a view to determining antibiotic sensitivity. The most likely bacterial culprits are Streptococcus equi sub. zooepidemicus while Escherichia coli, Pseudomonas aeruginosa, Klebsiella spp., Leptospira spp., Enterobacter spp., α-haemolytic Streptococci, Staphylococci spp. and nocardioforms have also been implicated.[7] The most common mode of access is via the cervix, which has been in some way compromised, with the pathogen then ascending up through the uterus – this form known as “ascending placentitis” accounts for over 90% of placentitis cases [8]. Antibiotic therapy owing to the difficulties of identifying sensitivities, is commonly broad-spectrum. Another important aspect of the antibiotic is that not all antibiotics have an ability to penetrate reproductive tissues and access allantoic/amniotic fluids to achieve minimum inhibitory concentrations (MIC). The TMP-SMZ group are often used, which provide reasonably broad coverage, but also good penetrative ability[9].
Both potentiated sulphonamides (trimethoprim sulfamethoxazole, 30 mg/kg, BID PO) and penicillin (procaine penicillin 22,000 IU/kg BID IM or crystalline penicillin 22,000 IU/kg QID IV) have been demonstrated to cross the placenta and consequentially may be valuable where organisms susceptible to these drugs are identified. [10][11][12] Gentamicin (6.6mg/kg SID IV) however, was not demonstrated to achieve a high enough concentration in the allantoic fluid to inhibit Streptococcus equi [13]. Despite that, it may achieve adequate concentrations to be effective against E. coli and Klebsiella spp. [10][11]
Douglas showed that placentitis mares treated with estrogen were significantly more likely to produce a live foal than affected mares that did not receive estrogen: 70% vs. 20% (n=20 and 50 respectively). Either estradiol cypionate (ECP) or estradiol 17β at a dose rate of 10 to 20 mg sid or bid was given intramuscularly.[5][14] In that same paper, the author comments that in his experience, mares presenting with an estrogen level of 300 pg/ml or lower had less than a 15% live foal rate irrespective of what therapy was initiated. More recent research has shown that when the “standard” treatment of antibiotics, Flunixin meglumine and Altrenogest (Regumate) are used, estrogen levels are lower, and therefore recommends supplementation with Estradiol Cypionate, observing that “inclusion of estradiol cypionate as part of the treatment for experimentally ascending placentitis appears to be more favorable than altrenogest”.[15]. This echoes the findings in separate research which observed “In conclusion, in addition to basic treatment TMS+FM [Trimethoprim Sulfa + Flunixin meglumine], mares with experimentally induced ascending placentitis benefited from ECP supplementation. Conversely, ALT [Altrenogest] did not appear to make a difference in outcomes”.[16] It is hypothesized that a promotion of the uterine immune response occurs from the additional estrogen, likely as a result of increasing uterine bloodflow, coupled with the fact that ECP is also positively pro-inflammatory, and will activate the specific immune cells/signaling molecules that are needed to properly attack a bacterial invasion. Another drug that is sometimes used in placentitis-affected mares with the intention of increasing bloodflow to the uterus and placenta is Isoxsuprine, the value of which is however not proven.
Reduction of Inflammation:
“Placentitis” means “inflammation of the placenta”, so obviously reduction of the inflammation and suppression of pro-inflammatory cytokines is going to be beneficial. This is typically initiated by use of a non-steroidal anti-inflammatory such as Flunixin Meglumine (e.g. brand names: “Banamine®“, “Finadyne®“). Other drugs that may be used with the intent of reducing inflammation include Pentoxifyline, Aspirin, and Phenylbutazone. The NSAIDs have the added advantage of being prostaglandin-release inhibitors, which can be advantageous in this situation, as PGF2α is the hormone that will be significantly responsible for the foaling (or abortion) process.
Encourage Myometrial Quiescence
The myometrium is a layer of the uterine wall that is predominantly smooth muscle with the main function of inducing uterine contractions. Clearly therefore, suppression of this activity if it occurs prior to the desired foaling point is preferable. Drugs that are going to be beneficial in suppressing these contractions are categorised as “tocolytics”. One easily available tocolytic that has in the past been used in the placentitis-affected mare is Clenbuterol (brand name “Ventipulmin®“). The ability of Clenbuterol to suppress smooth muscle contractions over a prolonged period of treatment however has been suggested as being minimal to nil [17], so the value in long-term placentitis treatment must be questioned. Of even more concern with the use of Clenbuterol is the research that indicates the ability of the drug to not only relax smooth muscle of the uterus but also the cervix, and that it actually has the potential to cause earlier delivery [11][12][18][19]. Clenbuterol therefore is not to be recommended for use in the placentitis-affected mare. A more important and commonly used drug to reduce myometrial activity is the oral progestin Altrenogest (brand name “Regumate®“, also available as an injectable compound from BET Pharm, Lexington, KY, USA). It should be noted that Altrenogest was found to increase endogenous levels of another progestin – 5αDHP – which is important in uterine quiescence. This was contrary to the effect of short-term treatment with P4 (Progesterone) given at 300 days of gestation, which not only did not increase 5αDHP, but actually shortened gestational duration. [20] It must however also be noted that other research has found that in mares with experimentally induced ascending placentitis Altrenogest treatment did not appear to make a difference in outcomes.[16] Concerns are often voiced that the use of Progestin therapy could result in the inappropriate maintenance of a compromised pregnancy. It has however been shown that mares will “foal through Regumate” [21]. Treatment of placentitis-affected mares with a double-dose of the oral version of altrenogest “Regumate®” has been suggested, [22][23] but while beneficial under the circumstances, the effect of this dosage on “forced” maintenance of an undesirable pregnancy has not been established.
Other Possible Treatments
It has been suggested that the treatment of the placentitis-affected mare with Dexamethasone could be beneficial in that it accelerates fetal maturity in the equine. Unlike in some other species – notably the bovine – Dexamethasone at a dose of up to 100 mg IM during late pregnancy did not cause abortion or delivery of a premature foal, but was shown to accelerate fetal maturity and shorted the gestational duration significantly. [24] This paper did however observe: “Dexamethasone stimulates precocious fetal maturation and delivery in healthy late pregnant mares. However, fetal HPA activity may be suppressed… Dexamethasone treatment could be used to improve foal viability in mares at risk of preterm delivery. The endocrine effects of such a therapy must be evaluated before clinical intervention with glucocorticoids can be recommended.“. Caution is therefore recommended before exploring this route of treatment in a clinical or farm setting. Interestingly, Gravett et. al. found that combined treatment which included Dexamethasone of non-human primates delayed onset of pre-term labour in those species.[25]
Duration of Treatments
The question of how long to continue treatments can result in some conflicting viewpoints. It would seem logical that treatments intended to maintain the pregnancy – rather than treat the placentitis condition – should not be maintained beyond a point where the foaling could result in a full-term foal. Exactly what that point is going to be is of course not known for the individual animal, only as a generality for the species as a whole. The general “rule of thumb” for full-sized horses is that it is unlikely that a foal will survive if born before 300 days; that between 300 and 320 days varying levels of neonatal intensive care may be required in order to maintain a foal – and the earlier in that time frame the foaling occurs, or the lesser the neonatal intensive care therapy, the lower will be the survival rate; by 320 days of gestation, one is entering the range of normal durations however individual animal conditions will vary and premature foals may still be seen. There is a further complication to the earlier of these prospective time frames in that a fetus that has been affected by a placentitis situation may actually “accelerate” maturity with the production of a live mature foal earlier than anticipated. As not all of the foal’s organs develop at the same rate, although earlier than normal delivery of a mature foal is a possibility, so too is the delivery of a compromised foal requiring intensive care. Induction of parturition in the placentitis-affected mare is not recommended, as final maturation of the fetus only occurs in the last 3-5 days before foaling and the external human is unable to predict that time point.[1] Use of common pre-foaling test indicators such as pH value milk-test strips are unreliable in these mares.
One approach is to cease treatment with drugs calculated to suppress myometrial activity – the tocolytic group – as well as the prostaglandin-suppressor drugs (the NSAID group) at a point calculated to allow a live full-term foal delivery. 310 days has been suggested as a suitable point, although this is likely variable between cases. Conversely, it is considered beneficial to maintain antibiotic and estrogen therapy through to term as these drugs are unlikely to interfere with the foaling process, but may remain beneficial in combating any uterine pathogenic presence. It has also been suggested that given the ability of the mare to “foal through” Altrenogest, that the treatment with that drug can be continued until foaling in order to maintain uterine quiescence.[1]
Prolonged antibiotic therapy of the mare is not without risk factors for the mare or the fetus. For example Sulfa drugs can create gastro-intestinal problems in the mare, while Gentamicin remains longer in the fetal compartment than in the mare’s circulatory levels and prolonged use can damage the foal’s kidneys.
It should be reinforced that individual circumstance dictate individual treatment periods. If a mare has undergone prolonged treatment during her pregnancy, then it may be acceptable to cease some treatments sooner rather than later – especially if there are indications of a reduction of placental inflammation per ultrasound. Conversely, a mare that has only recently commenced treatment is more likely to require continuation of the therapies closer to term.
Sequela and Follow-up
It is important to recognise that mares that have undergone placentitis during the current pregnancy are more likely to experience placenta previa – also known as a “red bag delivery” or “premature placental separation”. In this situation, instead of the allanotoic sac being ruptured during the foaling process – typically by the foal’s feet – with the subsequent release of the allantoic fluid (the “waters breaking”) and the passage of the foal within the amniotic sac, the entire (or a major portion of the) allantoic sac is presented externally in an unruptured state with the foal still fully enclosed. In the worst case scenario, with an unattended foaling, this will typically result in a dead foal. Availability of oxygen is beneficial in the case of placenta previa – with the mare receiving the oxygen during the foaling process and the oxygen source being immediately transferred to the foal once delivered. The hypothesis on the use in the mare is the intent to hyper-oxygenate the mare’s blood source, which may then increase potential oxygen transfer across the limited placental attachment. Obviously this will only be beneficial in the case of partial separation during foaling. The neonatal foal delivered in a planceta previa situation is almost certainly going to be hypoxic to some degree, with hypoxic ischemic encephalopathy (“HIE” – formerly known as “dummy foal syndrome” or “neonatal maladjustment”) being the issue. Increased oxygen supply during and immediately following foaling may assist in reversing some of the negative effects, although one should remain aware of the issue and be on guard for other symptoms. Hyperbaric oxygen chamber treatment has also been indicated as beneficial in the human and the equine neonate that has experienced HIE[26][27] however availability of such units suitable for the equine is limited.
Evaluation of the placenta following foaling can be useful to determine if the the placentitis was of an ascending form, which would suggest external causative pathogenic source, or if the inflammation is solely limited to the uterine horn region, which would suggest that the source originated from within the uterus. This determination, although not absolute when based upon placental evaluation, can be a useful guide to future management.
Another significant concern following delivery of the foal from the mare affected with placentitis is that the neonate is likely to have been exposed to the causative pathogen in utero. Consequently the foal must be considered at high risk for neonatal septicaemia. Immediate use of a course of broad-spectrum antibiotics administered by injection at birth is often considered desirable and a life-saver.
Following foaling, the mare should be considered for further treatment as despite having been treated during pregnancy, it is extremely likely that a uterine pathogen remains present. In particular if the mare is to be used for future breeding, it is important that treatment to clear that pathogenic presence be performed sooner rather than later after foaling. Consideration should also be given to how the causative pathogen gained access to the uterus during pregnancy. Clearly in the case of ascending placentitis there is a compromising of the cervix. Digital evaluation of the cervix prior to re-breeding should be performed during diestrus (when the mare is not in heat) looking for tears or scar tissue which may be a contributory factor.
If the mare is bred in the future, and a clear cause was not identified and corrected, it is thought to be potentially beneficial to place the mare on exogenous progestin therapy for the duration of the pregnancy. This is done with the intention of tightening the cervix to prevent pathogenic access (this may also be considered even if there was cervical damage that was repaired), as well as maintaining uterine quiescence. Another therapy that has not been scientifically evaluated, but which is often recommended is the prophylactic treatment of “at risk” mares with a broad-spectrum antibiotic for 7-10 days every month of pregnancy from the fourth month onwards. The hypothesis for the value of this treatment being an ongoing control of any uterine-accessing pathogen with any resulting infection if not being completely eliminated at least being kept to a “low murmur” level that does not result in full-blown placentitis.
How Successful is Treatment?
Clearly there will be case-dependant and individual factors that will affect outcome, so it is very difficult to put a standardised set of success statistics in place. In research that looked at bacterially-inoculated pregnant mares, there was a clearly definable difference between those mares in a treated group compared to untreated controls. The treated group – which received antibiotic (trimethoprim sulfamethoxazole), anti-inflammatory agent (pentoxifylline) and a synthetic progestin treatment (altrenogest) such as are outlined in the list above in this article – had a viable foal production rate of 83% (10/12). This contrasted sharply with the non-treated control group which had a 0% survival rate (0/5).[28] Although an experimentally-induced placentitis may not relate exactly to a naturally occurring case, this data does provide strong evidence that suitable treatment can significantly increase the likelihood of a successful outcome if that treatment is initiated at an early enough point. Immediacy of treatment is possibly going to be on one of the leading factors related to successful outcome (outside of course suitability), so delaying onset of diagnostics and treatment is strongly discouraged. Evaluation of the mare as outlined above should be performed as soon as an irregularity is identified. With placentitis, the old adage “better safe than sorry” most definitely applies, and it is better to have the mare checked and find no problem, than to wait until there is no doubt that a problem exists.
Case Report
14 year old, 1,100 lb (500 kg) multiparous mare with a history of placenta previa related to placentitis. The previous year, with the placenta previa, the foal had been presented dead despite it being an attended foaling with opening of the placenta and rapid assisted delivery performed.
Timeline
- Day 304 mare presented with a full, tight udder. Fluid expressed was thick, honey coloured and salty. Under normal conditions mammary development at this stage – particularly with honey coloured salty secretions – may not be considered unusual, but as a consequence of the history, the mare was being closely monitored and evaluations were immediately performed upon this initial suggestion of a problem.
- Mare was ultrasounded, and an increased CTUP identified (although this may not be relevant given Löf et. al’s observations about CTUP measurements in the last 30 days of pregnancy – see above and reference [4]).
- Immediately placed on 220 mg (10 mls) of oral Altrenogest (Regumate®), 550 mg (11 ml) IV Flunixin Meglumine (Banamine®) and 15 grams of trimethoprim sulfamethoxazole twice a day, morning and evening. The mare was also injected every third day with 50 mg Biorelease Estradiol cypionate (ECP-LA Bet Pharm., Lexington, KY) IM.
- Day 305, udder was soft, flacid and fluid expressed was less viscous.
- By day 306, udder was again full and tight and mare was fully waxed. Fluid expressed was thick, viscous and sticky again.
- Day 307 udder was taught and fluid expressed was white. Tested 300 ppm calcium and 6.8 pH
- Treatment was continued. Calcium tested 300 to 400 ppm and pH tested 6.4 to 6.8 for the next 24 days, during which the mare would sporadically drip and squirt milk.
- Day 331, the mare foaled. Milk calcium level prior to foaling was 400 ppm and pH dropped to slightly below 6.4
- Partial placenta previa was observed during the foaling process, however the foal was markedly small for this mare and stallion combination, so the foaling process was very quick as well as aided.
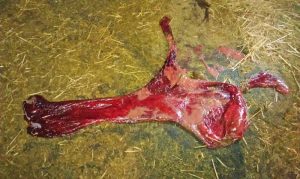
- The placenta when passed was found to be discoloured with significant pockets of pus present (see photos).
- The foal was immediately administered stored colostrum from a source other than the mare as well as prophylactic treatment with a Ceftiofur sodium antibiotic course for 5 days.
Case Comments
The close monitoring and prompt treatment quite likely saved this foal. It has been repeatedly shown that the earlier that treatment of the mare affected with placentitis is performed, the more likely there is to be a successful outcome. Notwithstanding the prompt onset of treatment once symptoms were seen, there was clearly compromising of placental function present long before diagnosis occurred. This is evidenced by the purulent material still present within the placenta despite antibiotic treatment for over 3 weeks, but more significantly by the distinctly reduced size of this warmblood foal out of a 16 hh (1.625 meters) mare and by a 16.2 hh (1.676 metres) stallion. Birth weight was estimated at no more than 60 lbs (27.2 kilos). Previous foals out of this mare or by this stallion have been “normal” sized for the type of animal – in the region of 100-120 lbs (45-54 kilos). The lack of definitive change in milk pH and Calcium levels between day 307 and the actual day of foaling clearly demonstrate the risk of relying on a milk sample reading to indicate imminent foaling in a placentitis-afflicted mare. The importance of being in attendance at foaling is increased owing to the risk of placenta previa.
“Click” on all images to enlarge
References:
1: LeBlanc MM. Placentitis: Important Facts. Unpublished
2: Troedsson MHT, Renaudin CD, Zent WW, Steiner JV. (1997) Transrectal Ultrasonography of the Placenta in Normal Mares and Mares with Pending Abortion: A Field Study. Proc. AAEP Annual Convention: 256-258
3: LeBlanc MM, Macpherson M, Sheerin P. (2004) Ascending Placentitis: What We Know About Pathophysiology, Diagnosis, and Treatment. Proc. 50th AAEP Annual Convention: 17-143
4: Löf HK, Gregory JW, Neves AP, Jobim MIM, Gregory RM, Rodrigo C. Mattos RC. (2014) Combined thickness of the uterus and placenta (CTUP) as indicator of placentitis in Thoroughbred mares. Pferdeheilkunde 30;1: 37-41
5: Douglas RH, Endocrine Diagnostics in the Broodmare: What you need to know about Progestins and Estrogens. Unpublished.
6: Coutinho da Silva MA, Canisso IF, MacPherson ML, Johnson AE, Divers TJ. (2013) Serum amyloid A concentration in healthy periparturient mares and mares with ascending placentitis. Equine Vet J. 45(5):619-24
7: Giles, R. C., Donahue, J. M., Hong, C. B., Tuttle, P. A., Petrites-Murphy, M. B., Poonacha, K. B., Roberts, A. W., Tramontin, R. R., Smith, B., Swerczek, T. W.(1993). Causes of abortion, stillbirth, and perinatal death in horses: 3,527 cases (1986-1991). J Am Vet Med Assoc 203: 1170-1175
8: Whitwell KE. Infective placentitis in the mare. (1988) In: Powell DG, ed. Equine infectious diseases V: Proc. of the 5th International Conference. Lexington, KY: University Press of Kentucky 172-180
9: Sertich PL, Vaala WE. (1992) Concentrations of antibiotics in mares, foals and fetal fluids after antibiotic administration in late pregnancy; Proc. 38th AAEP Annual Convention: 727-736
10: Murchie TA, Macpherson M, LeBlanc MM, Luznar S, Vickroy TW. (2003) A microdialysis model to detect drugs in the allantoic fluid of pregnant pony mares. Proc. 49th Annual Convention AAEP
11: Macpherson ML. (2005) Treatment strategies for mares with placentitis. Theriogenology. 64:528-534
12: Troedsson MH, Macpherson ML. Diagnosis and treatment of equine placentitis. Proceedings of the XXVIII Congreso Anual. Mexico. 2006
13: Santschi, E. M. and Papich, M. G.(2000). Pharmacokinetics of gentamicin in mares in late pregnancy and early lactation. J Vet Pharmacol Ther 23: 359-363
14: Douglas RH. 2004 Proc. Soc. Theriogenology. Invited Paper
15: Curcio BR, Nogueira CEW, Pazinato FM, Borba LA, Feijo LS, Scalco R, Canisso IF. 2018. Plasma estradiol-17β Concentrations in Mares Treated for Experimentally Induced Ascending Placentitis. Theriogenology 66:243
16: Curcio BR, Canisso IF, Pazinato FM, Borba LA, Feijó LS, Muller V, Finger IS, Toribio RE, Nogueira CEW. 2017. Estradiol cypionate aided treatment for experimentally induced ascending placentitis in mares. Theriogenology 15;102:98-107
17: LeBlanc M.M., Personal communication
18: Card CE, Wood MR. (1995) Effects of acute administration of clenbuterol on uterine tone and equine fetal and maternal heart rates. Biol Reprod Monogr. 1:7-11
19: Palmer E, Chauvatte-Palmer P, Duchamp G, Levy I. (2002) Lack of effect of clenbuterol for delaying parturition in late pregnant mares. Theriogenology. 58:797-799
20: Ousey JC, Rossdale PD, Palmer L, Houghton E, Grainger L, Fowden AL. (2002) Effects of progesterone administration to mares during late gestation. Theriogenology 58:793-795
21: Shoemaker CF, Squires EL, Shideler RK. (1989) Safety of altrenogest in pregnant mares and on the health and development of offspring. Equine Vet. Sci. 9:69-72
22: LeBlanc MM. (2004) Infertility: Mares that conceive and lose their pregnancy. Proc.of the 10 Congresso Nazionala Multisala SIVE. Perugia
23: Ousey JC. (2004) Peripartal endocrinology in the mare and foetus. Reprod Domest Anim. 39:222-231
24: Ousey JC, Kölling M, Kindahl H, Allen WR. (2011) Maternal dexamethasone treatment in late gestation induces precocious fetal maturation and delivery in healthy Thoroughbred mares. Equine Vet J. 43(4):424-9
25: Gravett, MG, Adams KM, Sadowsky DW, Grosvenor AR, Witkin SS, Axthelm MK, et al. (2007) Immunomodulators plus antibiotics delay preterm delivery after experimental intraamniotic infection in a nonhuman primate model. Am J Obstet Gynecol 197(5):518
26: Zhou BY, Lu GJ, Huang YQ, Ye ZZ, Han YK. (2008) Efficacy of hyperbaric oxygen therapy under different pressures on neonatal hypoxic-ischemic encephalopathy. Zhongguo Dang Dai Er Ke Za Zhi. 10(2):133-5. (Article in Chinese)
27: Slovis N. Hyperbaric oxygen therapy in horses. Proc ACVIM 2006;150-152
28: Bailey CS, Macpherson ML, Pozor MA, Troedsson MH, Benson S, Giguere S, Sanchez LC, Leblanc MM, Vickroy TW. (2010) Treatment efficacy of trimethoprim sulfamethoxazole, pentoxifylline and altrenogest in experimentally induced equine placentitis. Theriogenology 74(3):402-12
Our thanks to Avalon Equine for the case history and photographs of the placentitis case. Also our thanks to Dr. Jonathan Pycock, B. Vet. Med., PhD, D.F.S.M, M.R.C.V.S. for permission to use his ultrasound images of CTUP measurement.
© 2014 & 2019, Equine-Reproduction.com, LLC
Use of article permitted only upon receipt of required permission and with necessary accreditation.
Please contact us for further details of article use requirements. Other conditions may apply.