Ultrasound Characteristics of the Uterus in the Cycling Mare and their Correlation with Steroid Hormones and Timing of Ovulation
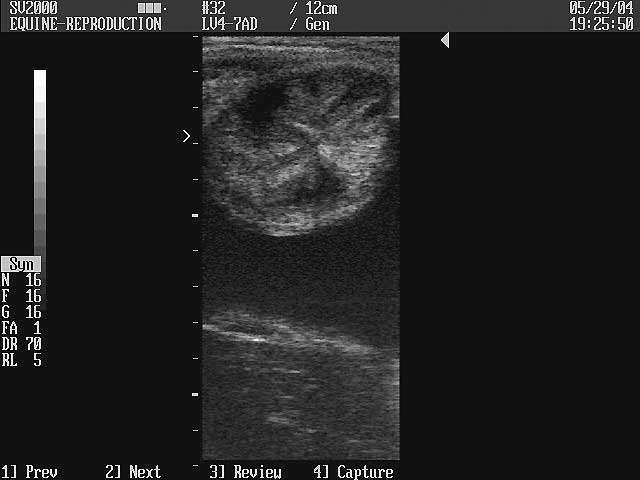
The introduction of transrectal ultrasound technology to visualise the reproductive tract in mares (Palmer and Driancourt 1980) has allowed cyclical changes in the ultrasonic morphology of the reproductive tract to be studied. One of the most striking characteristics as visualised using ultrasonography is the appearance of the endometrium during the oestrus cycle. During dioestrus, individual endometrial folds are not visible and the uterus has a homogenous echotexture. The lumen of the body of the uterus is often visible as a white line formed by specular reflections at the closely apposed lumenal surfaces in marked contrast to the situation during oestrus when individual endometrial folds can be visualised (Ginther and Pierson 1984). It is thought that endometrial folds become oedematous during oestrus due to increased concentrations of circulating oestrogen (Hayes et al 1985). This gives a heterogenous image with the dense central portions of the folds appearing echogenic and the oedematous portion of the fold represented by non-echogenic areas.
When the uterine horn is imaged as a cross-section, the appearance resembles a sliced-orange or cartwheel. Occasionally free oestrus fluid can be imaged. It is possible to grade the degree of endometrial detected and this concept was introduced by Ginther and Pierson (1984) and later developed by Squires et al (1988) and Allen (1989). The scores of endometrial oedema have been found to reflect changes in circulating oestrogen and progesterone concentrations and sexual behaviour scores (Hayes et al 1985). Endometrial folds first become visible at the end of dioestrus, become more prominent as oestrus progresses and generally diminish from approximately 2 days from ovulation until the day of ovulation when oedema may have disappeared. This is supported by three studies which used thoroughbred and standardbred or pony type mares (Ginther and Pierson 1984; McKinnon et al 1987; Allen 1989). However, one recent study using Belgian draft mares found that the prominence of endometrial folds peaked at 6.6 days before ovulation and abruptly veterinarians dealing with different type of mares.
The mare has an oestrous period that lasts, on average, between 5 and 7 days. For pregnancy rates to be maximal, normal mares should be bred within 48 hours of ovulation. For mares bred artificially, insemination should be even closer to ovulation. There are a wide range of parameters used to estimate the time of ovulation. These include follicle size, softness, and shape. The interpretation of endometrial ultrasonic morphology also forms an important part of establishing an accurate estimate of the stage of the oestrus cycle.
The purpose of the present study was to investigate the precise relationship of endometrial oedema to oestrous behaviour, stage of the oestrus cycle and plasma oestradiol and progesterone concentrations.
The following specific hypothesis was also investigated: Is the degree of ultrasonically visible endometrial oedema directly related to plasma oestradiol levels and does it only occur when progesterone levels are basal, or is oedema only related to the day of the cycle? Previous studies have investigated either oedema scores or oestradiol and progesterone concentrations, but this was thought to be the first study which combined all aspects.
Circulating concentrations of oestrogen and progesterone have been well documented for the oestrus cycle of the mare and recently reviewed by Ginther (1993a). Many of the rhythmic changes in the reproductive tract of the mare are under the control of the ovarian steroids. These changes during oestrus and dioestrus are due to the influence of the ovarian steroids that predominate during a given phase of the cycle (oestrogens and progesterone respectively). The period known as oestrus during which the mare is sexually receptive to the stallion is often determined by exposing the mare to a stallion (teasing).
In mares, oestradiol is thought to be responsible for oestrous behaviour (Nett 1993) and oestrogen concentrations during oestrus correlate well with sexual behaviour and grossly observable changes in the reproductive tract. Progesterone, on the other hand, inhibits oestrus behaviour and during oestrus, progesterone concentrations in plasma are below 1 ng/ml (Ginther 1993a). The effects of progesterone on behaviour and morphologic characteristics of the uterus are reported to be dominant over the effects of oestrogen (Daels and Hughes 1993).
MATERIALS AND METHODS
Mares:
Five Dutch warmblood mares with normal reproductive histories and weighing 600 to 750 Kg were used. The age of the mares ranged from 5-15 years and the examinations took place during the period March to May. All mares were non-lactating and in good general health and body condition. None of the mares had any apparent ovarian or uterine abnormalities and were cycling. The mares were housed, fed corn and hay twice daily and had access to grass pasture at least once per day.
Clinical and Laboratory Findings:
| Aggressive to the teaser: | 0 |
| Indifferent to the teaser: | 1 |
| Stands with the teaser and everts clitoris (winking): | 2 |
| Stands with the teaser, winking and passing urine: | 3 |
Gynaecological examinations including ultrasonic echography:
The genital tract was evaluated daily by both palpation and real-time, B-mode ultrasonography using a 7.5 MHz linear array transducer. The diameter and location of all follicles greater than 20 mm and all luteal structures was noted, but this data is not presented here. Ovulation was diagnosed by the disappearance of the follicle and the presence of an early corpus luteum (CL) as determined by both palpation and ultrasonic imaging.
Length of the oestrous cycle (interovulatory interval) was defined as the number of days between ovulations. The day of ovulation was designated Day 0 and was the first day that a large follicle observed the previous day was no longer present and a corpus luteum was detectable. Dioestrous ovulations were not used to calculate interovulatory intervals. Where the mare had a double ovulation during oestrus, the interovulatory interval was based on the first ovulation of the set.
The uterine echotexture was graded based on the following scheme:
| No oedema with a typical homogeneous echotexture characteristic of dioestrus: | 0 |
| Smallest amount of readily detectable uterine oedema, no free fluid: | 1 |
| Moderate amount of oedema, throughout the whole uterus: | 2 |
| Most obvious oedema throughout the whole uterus, sometimes free fluid also noted: | 3 |
Blood sampling:
Heparinised blood samples were collected from the jugular vein once daily at the same time throughout the experiment. They were centrifuged for 15 minutes and the centrifuged plasma stored at -20°C until assayed. Progesterone and oestradiol-17β concentrations were estimated by radioimmunoassay.
RESULTS
Overall, 12 oestrous cycles for the five mares were monitored. The length of the oestrous cycle was 23.5 (+ 1.9) days, with a dioestrus period of 16.5 + 1.1 days and an oestrus period of 7.0 (+1.1) days. The oestrus periods were all of sufficient length to allow normal oedema development i.e. more than five days. One mare had a double ovulation with the second ovulation occurring with 24 hours of the first.
Plasma oestradiol and progesterone concentrations: In all of the cycles, plasma oestradiol concentrations began to increase steadily during oestrus, beginning 5 to 7 days before ovulation. Peak concentrations were reached 24 to 48 hours before ovulation. Concentrations were rapidly decreasing at ovulation and basal dioestrous values reached within 48 hours of ovulation. For all oestrous cycles in this study, this sharp peak in oestradiol concentrations was noted.
Plasma progesterone concentrations also showed a similar pattern for the cycles of all the mares in this study. During oestrus, plasma concentrations were below 0.5 ng/ml. immediately after ovulation, concentrations rapidly increased to reach peak dioestrous values 5 to 6 days after ovulation. Values remained high throughout dioestrus, with a rapid decline beginning 14 to 15 days after ovulation.
Endometrial oedema and oestrous behaviour: Endometrial oedema was detected at some point during oestrus for every cycle investigated in this study. Endometrial oedema of score 0, 1, 2 or 3 was detected in 0, 17, 75 and 8% of mares, respectively, on day -3 (Day 0 = ovulation); 0, 8, 50 and 42% of mares, respectively, on day -2; 8, 26, 58 and 8% of mares, respectively, on day -1; 16, 58, 26 and 0% of mares, respectively, on day 0; 66, 26, 8 and 0% of mares, respectively, on day +1 and 83, 17, 0 and 0% of mares, respectively, on day +2.
For the twelve cycles as a whole, endometrial oedema first appeared 7.4 + 1.1 days before ovulation and peaked 24 to 48 hours before ovulation. In all but one of the 12 cycles, there was detectable oedema on the day of ovulation. However, the oedema on the day of ovulation was always less than at some point earlier during oestrus and, in any case, oedema never persisted more than one day after ovulation.
The appearance of oedema preceded the beginning of oestrous behaviour by 1, 2 or 3 days, although oestrous behaviour generally persisted 1 day after oedema disappeared.
Relationship between endometrial oedema, progesterone and oestradiol concentrations: Oedema was associated with basal (<1.0 ng/ml) progesterone values on all but one occasion. On that occasion, the oedema was grade 1 only. No oedema was ever detected with a progesterone concentration above 3 ng/ml.
There was no obvious direct relationship between oedema and oestradiol concentrations. Furthermore, within the same cycle, a mare often had the same oedema score early in oestrus as she had with double the oestradiol concentration two days later in oestrus. Some mares had consistently more oedema than others despite lower oestradiol concentrations.
DISCUSSION
The interovulatory interval in the Dutch Warmblood mares described here was similar in length to that described for other breeds of horses by Ginther (1993). The slightly longer period noted here could be due to the timing of the observations relatively early in the breeding season although it should be noted that all mares had an ovulation within the month preceding the beginning of this study.
Similar to earlier reports, the ultrasonic anatomy of the uterus was affected by the stage of the oestrous cycle (Ginther and Pierson 1984; Hayes et al 1985; McKinnon et al 1987; Plata-Madrid et al 1994). The pattern of endometrial oedema was similar to that described in the pony mares and light horse (Appaloosas, quarter horses and Standard-breds) mares studied by Ginther and Pierson (1984), Hayes et al (1985) and McKinnon et al (1987) with the oedema beginning 7 or 8 days prior to ovulation, peaking 24-48 hours before ovulation and declining to a dioestrous appearance within 2 days of ovulation. One difference was that oedema was detected more frequently on both the day preceding ovulation and the day of ovulation than in other studies. This could be a genuine fdifference in the warmblood, but could also reflect the use of a 7.5 MHz frequency ultrasound transducer which gives an image with much greater resolution. This point was acknowledged earlier by Ginther and Pierson (1984).
As the Warmblood is a relatively heavier breed of horse, the results may have been expected to have been intermediate between these earlier studies and the more recent study of Plata-Madrid et al (1984) which used a heavy Belgian draft breed of horse. However, this was not the case with this early regression of endometrial folds not being found.
Endometrial behaviour was temporally related to behavioural signs of oestrus. It would, therefore, seem reasonable to attribute oedema to increasing circulating oestrogen concentrations. However, it is known that non-hormonal influences (endometrial swabbing, mating) can stimulate uterine oedema (Pycock, J.F., unpublished observation). Nonetheless, there was no uterine interference in the mares in this study and so it seems reasonable to conclude that oestrogen concentrations, as has been shown for ovarian activity, sexual receptivity and other grossly observable changes in the reproductive tract (Daels and Hughes 1993) are correlated with oedema. However, there was no direct correlation between oedema intensity and oestradiol levels. Munro, Renton and Butcher (1979) reported that the intensity of oestrus behaviour was not related to plasma levels of oestrogens although Nelson et al (1985) did report an association of silent oestrus with lowered oestradiol concentrations. The situation was much clearer in respect of plasma progesterone levels: oedema only occurred when levels were below 1 ng/ml and so it seems likely that progesterone is the dominant hormone with respect to endometrial oedema intensity as is the case with behavioural characteristics and morphological changes in the cervix and uterus (Daels and Hughes 1993).
The results of these experiments demonstrated that detection of endometrial oedema has a role to play assessing the stage of the oestrous cycle of the warmblood mare and, in turn, assisting in the decision on the optimal time on when to mate the mare.
Endometrial oedema is also useful to detect during the transitional `spring’ estrus. Pressure to breed mares early in the year before the onset of their natural breeding season can cause problems for the veterinarian. Because of the considerable variation in the duration of estrus during the transitional period, efficient breeding of the mare can be difficult. During the transitional period the behaviour is variable, ranging from total rejection of the stallion, to interest but resistance to him mounting, to normal acceptance. These behavioural signs can be consistent or inconsistent. It is recommended that the interval between matings should not exceed 2 or 3 days, although there have been no critical studies on the survival time of sperm in the mares’ genital tract. It is important not to begin breeding too early or this will result in the mare being mated many times. The appearance of uterine oedema is an indication that the follicle should ovulate within a few days. A key factor in the emergence out of vernal transition is the development of steroidogenic competence by the follicle leading to an increase in circulating oestrogen concentrations which cause the release of LH from the pituitary due to a positive feedback mechanism. Oestrogen is responsible for the appearance of uterine oedema (in the absence of progesterone) and so this may be why the detection of uterine oedema is important in signalling the emergence of the mare from the transitional period. The diagnosis is by thorough ultrasonic examination and rectal palpation which reveals transitional follicles reaching a preovulatory size of > 30 mm. Visual identification of a corpus luteum or progesterone levels above 4 ng/ml confirms that the first ovulation has occurred and hence the onset of normal ovarian cyclical activity. The treatment of mares in transitional stage is based on progesterone or progestogens, with or without the addition of oestradiol esters, involving several parenteral routes of administration. Progesterone can be administered as an oil-based intramuscular injection, orally as the synthetic progestogen altrenogest (Equine Regumate) or by using a progesterone-releasing intravaginal device.
REFERENCES
Daels, P.F. and Hughes, J.P. (1993) The normal oestrous cycle. In: Equine reproduction. Eds. A.O. McKinnon and J.L. Voss. Philadelphia, Lea and Febiger. pp 121-133.
Ginther, O.J. (1993a) Endocrinology of the ovulatory season. In: Reproductive biology of the mare, 2nd edition. Wisconsin, Equiservices. pp 233-290.
Ginther, O.J. (1993b) Characteristics of the ovulatory season. In: Reproductive biology of the mare, 2nd edition. Wisconsin, Equiservices. pp 173-233.
Ginther, O.J. and Pierson, R.A. (1994) Ultrasonic anatomy and pathology of the equine uterus. Therio. 2, 505-516.
Hayes, K.E.N., Pierson, R.A., Scraba, S.T. and Ginther, O.J. (1985) Effects of oestrous cycle and season on ultrasonic uterine anatomy in mares. Therio. 24, 465-477.
McKinnon, A.O., Squires, E.L., Carnevale, E.M., Harrison, L.A., Frantz, D.D., McChesney, A.E. and Shideler, R.K. (1988) Diagnostic ultrasonography of uterine pathology in the mare. Proc. 33rd
Ann. Conv. Am. Ass. equine Pract. pp 605-622.
Munro, C.D., Renton, J.P. and Butcher, R. (1979) The control of oestrous behaviour in the mare. J.Reprod. Fert., Suppl. 27, 217-227.
Nelson, E.M., Kiefer, B.L., Roser, J.F. and Evans, J.W. (1985) Serum oestradiol-17 concentrations during spontaneous silent oestrus and after prostaglandin treatment in the mare. Therio. 23,
241-262.
Nett, T.M. (1993) Oestrogens. In: Equine reproduction. Eds. A.O. McKinnon and J.L. Voss. Philadelphia, Lea and Febiger. pp 65-68.
Palmer, E. and Driancourt, M. (1980) Use of ultrasonic echography in equine gynaecology. Therio. 13, 203-216.
Plata-Madrid, H., Youngquist, R.S., Murphy, C.N., Bennett-Wimbush, K., Braun, W.F. and Loch, W.E. (1994) Ultrasonographic characteristics of the follicular and uterine dynamics in Belgian mares.
J. Eq. Vet. Sci., 14, 421-423.
Squires, E.L., McKinnon, A.O. and Shideler, R.K. (1988) Use of ultrasonography in reproductive management of mares. Therio. 29, 55-70.
Summary:
Ultrasound Characteristics of the Uterus in the Cycling Mare and Their Correlation with Steroid Hormones and Timing of Ovulation
Dr. Jonathan F Pycock, B.Vet.Med., Ph.D., D.E.S.M., M.R.C.V.S.
Equine Reproductive Services, Messenger Farm, Ryton, Yorkshire
Changes in the endometrial oedema pattern during the oestrous cycle of Dutch warmblood mares were evaluated using transrectal, linear-array ultrasonography. The uterus was examined each day and the degree of oedema scored. Peripheral blood samples were taken daily for measurement of plasma oestradiol and progesterone concentrations. Endometrial folds first appeared 7 days before ovulation and peaked 24 to 48 hours before ovulation. Oedema score decreased before ovulation, but there was almost always detectable ovulation on the day of ovulation. There was no direct correlation between oedema and oestradiol levels, but oedema was not detected with progesterone above basal levels. Evaluation of endometrial oedema would seem to be a useful part of the routine pre-mating examination of mares in indicating the optimal time to mate the mare and providing an instant indication of basal levels of progesterone.
The ultrasonographic appearance of the endometrium of the mare is variable depending on the stage of the oestrus cycle and associated changes in circulating concentrations of oestrogen and progesterone. However, the precise relationship of oestrogen and progesterone concentrations to the ultrasonographic detection of endometrial oedema is not clear. If the degree of oedema can be used as a guide to the relative concentrations of oestrogen and progesterone, this would enhance its importance in clinical practice. The purpose of this paper is to consider both the ultrasonographic development of endometrial oedema in detail and its usefulness as a clinical tool. The uterus of all mares was examined using ultrasound and the degree of oedema graded from 0 (no oedema) to 3 (obvious oedema). The oedema score was correlated with other clinical findings (oestrous behaviour, cervix relaxation and follicular development). In addition to a large number of mares in clinical practice, 5 mares were examined daily and also bled for estimation of circulating progesterone and oestradiol concentrations. Overall, 18 oestrous cycles for the 5 mares were monitored.
Oedema could be satisfactorily graded using this system. The outer areas of the folds were hyperechoic and the oedema was most marked in the inner areas of folds, corresponding with the stratum spongiosum. The degree of oedema was useful in assisting the prediction of the time of ovulation: scores generally decline 24 hours before ovulation, however in most cases there was still detectable oedema at the time of ovulation. Oedema only occurred with basal progesterone levels and this may have a significance in relation to early embryonic death.
In conclusion, endometrial oedema is useful for helping to establish the stage of the oestrous cycle and as an indicator of impending early embryonic death.