Is it Possible to Cause Earlier Regression of the Endometrial Cups to Permit a Return to Breedable Estrus in the Mare?
Unique to the early equine pregnancy is the invasive trophoblast of the chorionic girdle and its formation of the endometrial cup cells which secrete equine chorionic gonadotropin (eCG). This invasion occurs between days 35–38 after ovulation. The endometrial cups first become visible on the surface of the endometrium in a circular formation around the conceptus which will have earlier “fixed” and be typically located at the base of what will be known as the gravid uterine horn. Secretion of the hormone equine chorionic gonadotropin, will continue in most cases until eCG becomes undetectable in the mare’s blood between about 120-150 days after conception. The eCG promotes production of secondary follicles which will then undergo a luteinization process – either regular ovulation, or a process similar to development of the luteinized unruptured follicle which is seen at times when attempting to breed the mare, and is placed under the collective generic misnomer of “anovulatory hemorrhagic follicle” – to become secondary Corpora Lutea. These in turn secrete progesterone which will assist in pregnancy maintenance until other progestins secreted by the fetoplacental unit take over the function starting between days 60-80. If the mare loses the pregnancy after the commencement of eCG secretion and prior to the regression of the endometrial cups – and therefore cessation of the eCG secretion – she will not return to estrus and be likely to conceive until eCG levels have dropped below 1.4 IU/mL[1]. Depending upon the point in the breeding season when this occurs, it may preclude the practicality of rebreeding the mare in the same year. For breeding management purposes therefore, the ability to cause regression of the endometrial cups would be important, but has hitherto not been achieved. While prostaglandin treatment of the mare in this time frame may cause loss of functionality of the secondary CLs, it does not impact the endometrial cups and permit the mare to return to a successfully breedable state.
As early as 1954, Clegg et al. suggested that natural regression of the endometrial cups was caused by a maternal cell mediated immune response[2], and this was supported in subsequent research[3][4]. The current authors, Fedorka et al., indicate that the regression of the endometrial cups is governed by IFNγ-producing lymphocytes, and importantly, that they had observed an increase in IFNγ following the use of a commercially-available Mycobacterium cell wall fraction (MCWF) immunostimulant (Settle: NovaVive, Napanee, ON, Canada) in the postpartum mare. These findings led to the current research.

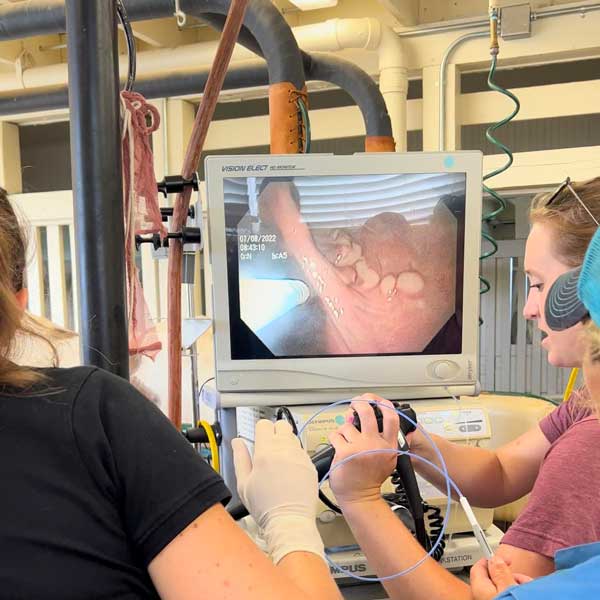
Videoscopic view of an endometrial cup pre- and post-injection with MCWF
(Image courtesy Dr. Fedorka)
Sixteen mares which had undergone an abortive process were divided into two groups. One group (10 mares) were to receive the MCWF treatment, while the remaining 6 mares acted as the control group, receiving no such treatment. A “treatment solution” was made of 6 ml MCWF combined with 14 ml Lactated Ringer’s Solution, to make a 20 ml total volume. Following ultrasound evaluation between what would have been 45-50 days of gestation, the treated group of mares received a Hysteroscopic-guided injection of 1 ml of the treatment solution into each visible endometrial cup, with any remaining volume being placed peripherally. This entire process was repeated 7 days later.
In the treated groups, seven days after treatment, a statistically-significant drop in circulating eCG levels was observed. This suppression remained evident over the next 35 days of evaluation. A higher percentage of treated mares (8/10) returned to estrus within a month of treatment, compared to 2/6 of the control mare group, a pre-ovulatory follicle being identified 23 days (+/- 4 days) in the responsive mares in the treated group. The treatment with the MCWF, which was shown to elevate IFNγ in these mares, was demonstrated to promote an earlier regression of the endometrial cups.
Our observation: The ability to cause earlier regression of the endometrial cups in mares which have undergone pregnancy loss is of obvious value if the mare is to be bred again within the same breeding season. In work done by the authors and others related to the paper, but not presented within it, at least 17 such mares have been treated in Lexington (KY) clinics with 12 successfully becoming pregnant following the shorter interval for regression of the endometrial cups (the remaining 5 have not had their pregnancy status reported to the researchers). Currently, the need for Hysteroscopic-guided treatment does limit the availability for general clinical use to more specific reproductive clinics, however this is minimally limiting in many countries. We suspect that further investigation may be performed to see if other routes of administration may achieve similar results, although the comparatively high dosages required associated with previously identified side-effects (transient elevated temperature) may present an inability to achieve this.
(Fedorka C, Schnobrich M, Muderspach N, Scoggin K, Dedman M, Weigle K, May M, Twist H, Linse C, Douglas R, Troedsson M. 2023. Utilizing an immunostimulant (MCWF; Settle) to hasten the degradation of endometrial cups. JEVS 125:104802)
References:
1: Estradé MJ, Cazales N, Perdigón F, Cavestany D, Mattos RC. 2016. eCG Concentration and Subsequent Reproductive Activity in Mares After Abortion at Day 70. JEVS 42:88-93
2: Clegg MT, Boda JM, Cole HH. 1954. The endometrial cups and allanto-chorionic pouches in the mare with emphasis on the source of equine gonadotrophin. Endocrinol. 54, 448-463
3: Amoroso EC. 1955. Endocrinology of pregnancy. Br. med. Bull. 11, 117-125
4: Allen WR. 1975. The influence of fetal genotype upon endometrial cup development and PMSG and progestagen production in equids. J. Reprod. Fert., Suppl. 23, 405-413.